The new technology can be applied to all foil types, matt or gloss. Different text patterns can be tested with one test method, making it ideal for production lines with multiple language print variations. The system is designed to allow for easy calibration, quick set up times and a streamlined validation process across different foil types.
The VisionScan 3D offers pharmaceutical manufacturers a flexible, reliable, deterministic and cost saving alternative to a destructive leak test for blisters. The method does not affect the integrity of the blister pack, allowing packs that have passed the test to be returned onto the production line.
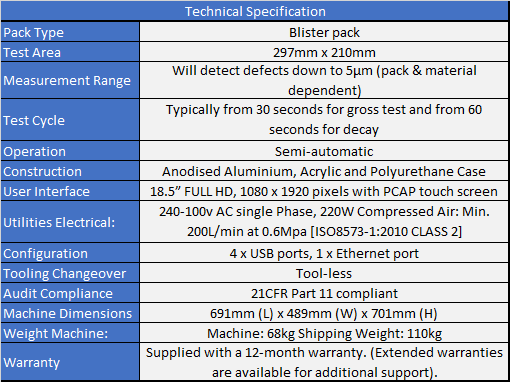
· Non-destructive, deterministic blister leak test device designed to test all foil types
· Incorporates patented 3D technology that can detect leaks in individual blister pockets, channel leaks and weak seals down to a 5μm laser drilled pin hole (pack and material dependent)
· Tool-less device making it ideal for production lines running multiple products
· Large test area (297x210mm) provides high throughput
· Can test multiple packs per test cycle
· Can test packs that contain tablets / capsules in multiple material / design formats
· Streamlined validation process for different foil types
· Rapid test time down to 60 seconds for micron holes and as low as 30 seconds for gross holes
· Simple operator use via a touch screen interface
· Operating system can store unlimited product types
· Network connectivity to a central server
· Can form part of 21CFR part 11 compliant system
· Capable of storing and exporting data for audit and quality control purposes
· Active Directory and flexible reporting built-in